Brain Structures and Outcome
In some hospitals, magnetic resonance imaging (MRI) is becoming an essential approach when aiming to identify what is "wrong" with the brain. This is particularly true in stroke patients, as it has been shown that stroke lesions and ischemia can be easily visible within the brain. With the advancement of MRI, additional disease within the brain may become visible. This offers the unique opportunity to investigate how these structures influence stroke outcome.
However, this is not only true for diseased tissue within the brain. MRI also allows us to quantify other brain structures that might be visible on these scans, opening up an entire field of research which aims to help doctors quickly gather all the information they might need to treat a patient. Before utilizing new information for treatment, however, we first need to identify how differences in these structures actually affect outcome.
White matter hyperintensity
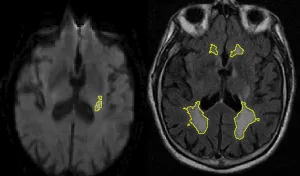
White matter hyperintensities (WMH) are a common disease of the brain's white matter and are considered to result from underlying small-vessel disease. Over the life-span, many people develop these hyperintense patterns, and it has previously been shown that stroke patients often exhibit an increased disease burden. As part of the ARTEMIS project, we developed a fully automated pipeline which allows us to identify diseased tissue and quantify the disease burden.
When the WMH is outlined, we can start asking more sophisticated questions. In terms of volume, many questions have been asked (and answered), however, summarizing a disease which clearly demonstrates spatial patterns (some areas are more prone to WMH than others), may not help us to fully understand disease progression and differences between patients. By utilizing the GASROS study, we were able to combine the disease information of an entire cohort into a single spatial prevalence map using the by us developed template, as it can be seen in the figure below.
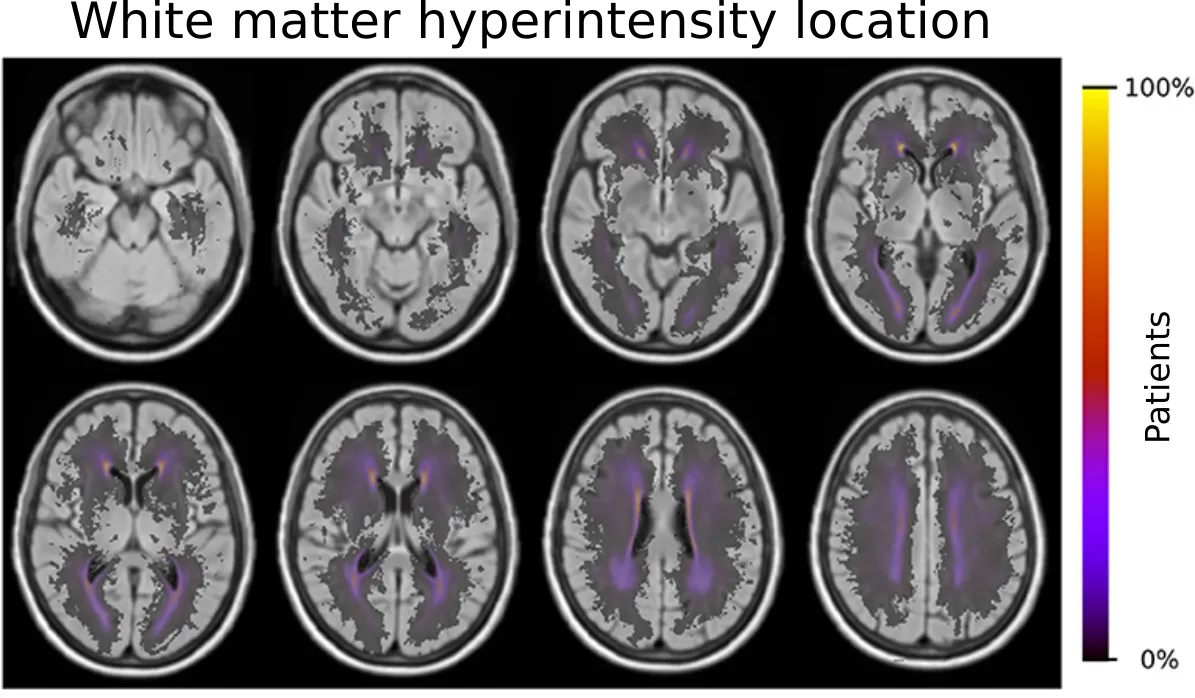
Importantly, the brain can be spatial differentiated into vascular territories based on their blood supplying arteries. To further our understanding of the spatial WMH patterns, we created a template to analyze these disease patterns based on their territories in stroke patients. This template, which allows for automated analyses of large scale data sets, is publicly available on Zenodo ("Vascular Territory template and atlases in MNI space").
In our investigation ("Spatial Signature of White Matter Hyperintensities in Stroke Patients"), we were able to demonstrate that older age, male sex, small-vessel stroke subtype, hypertension, hyperlipidemia, and smoking shifted the prevalence of the disease burden in these territories. This further highlights the importance of incorporating spatial information in any type of analysis, when it comes to disease burden that are not uniform.
Ventricles
Ventricles within the brain are spaces that are filled with cerebrospinal fluid. They serve, among others, to clean out toxins and other harmful things within the brain. In addition, their central location make them one of the most promenent features visible on an MRI examination. In general, ventricular volume increases with age, however, in certain diseases, the ventricles can become enlarged. Nonetheless, the ventricles have not been fully studied as a potential biomarker for outcome in stroke patients.
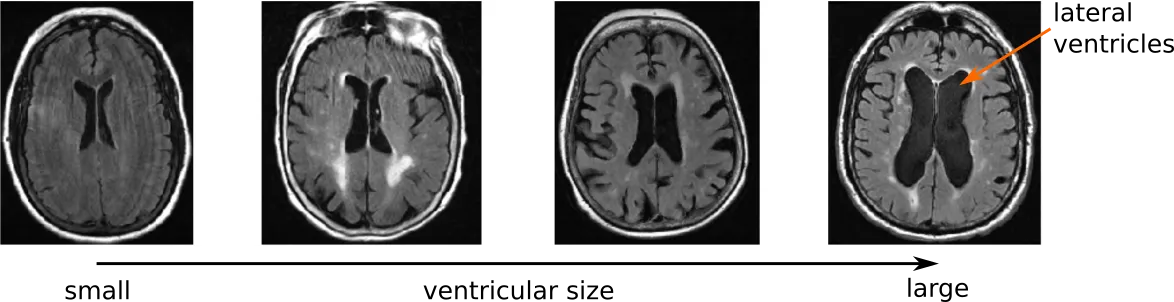
In order to study ventricles in stroke patients, we first developed an automated segmentation tool dedicated for clincal scans. This helped our efforts to understand stroke in two independent manners. First, we analyzed the shape of the ventricles. With this analysis, we were able to show that clinical aspects of stroke patients, such as hypertension, white matter hyperintensity volume, and outcome, are associated with specific shape differences (Patient-specific Conditional Joint Models of Shape, Image Features and Clinical Indicators), and is illustrated in the video below.
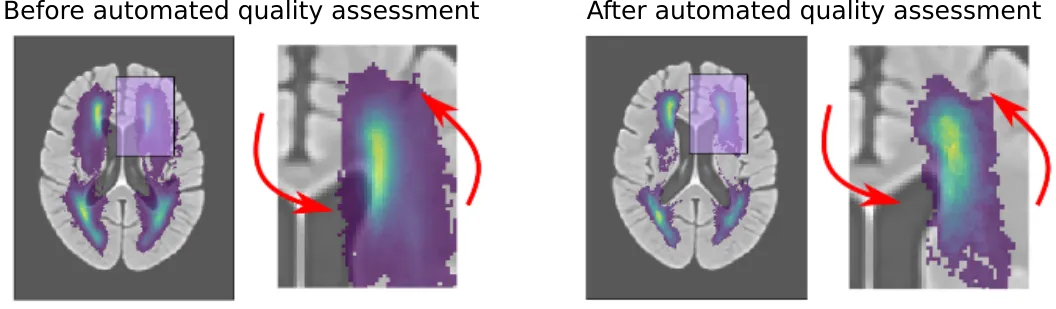
Additionally, the ventricle segmentation furthered our efforts in creating and evaluating automated image processing pipelines ("Automated Image Registration Quality Assessment Utilizing Deep-learning based Ventricle Extraction in Clinical Data"). Spatial analyses, as seen for example in the white matter hyperintensity section above, requires an image registration step, which principally overlays brains on top of a template. This is a difficult step, even for a computer. Things get expecially difficult when working with low resolution clinical data. In the clinic, and depending on the disease, MRI scans may have to be short due to not delay additional treatment.
While the registration can still be performed, it becomes important to assess if and how much the computer struggled with the alignment. Utilizing the ventricles, however, we were able to automatically assess the computers performance. With additional efforts to create age-specific templates as stepping stones, we were able to significantly reduce errors. This is further illustrated in the figure below.